Key Highlights
- Definium Therapeutics endorsed a White House Executive Order designed to fast-track mental health innovations, particularly psychedelic-based therapies
- The biotech firm is advancing DT120 ODT (lysergide tartrate), a novel LSD-derived formulation targeting GAD and MDD
- DT120 carries FDA Breakthrough Therapy Designation with four ongoing Phase 3 clinical trials
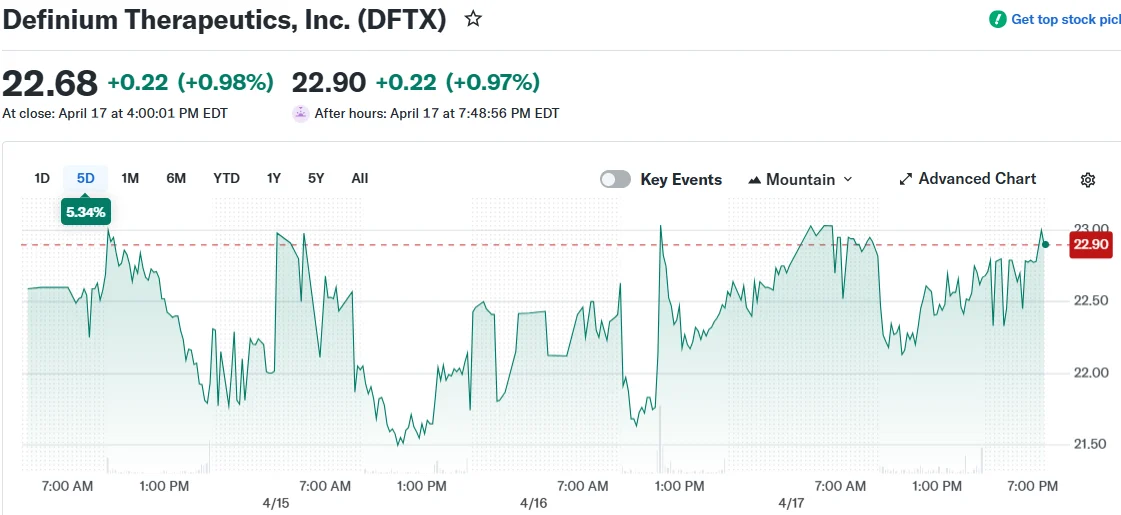
- On April 17, Stifel launched coverage with a Buy recommendation and $30 price objective, compared to current trading levels near $22.46
- Recent insider transactions reveal zero purchases alongside approximately $0.8M in stock sales during the previous quarter
On April 19, Definium Therapeutics (DFTX) released a public statement endorsing a fresh White House Executive Order designed to accelerate regulatory approval for psychedelic-assisted mental health interventions. Shares responded positively, climbing 0.98% following the announcement.
The presidential directive instructs federal departments to fast-track mental health treatment options, reduce bureaucratic barriers in the approval process, and foster greater interagency cooperation. Notably, the order explicitly identifies psychedelic compounds as promising therapeutic candidates for combating America’s escalating mental health emergency.
Rob Barrow, Definium’s Chief Executive Officer, praised the executive action as “an important recognition of the persistent unmet treatment needs in serious mental illness.” Barrow emphasized that the organization is progressing a comprehensive clinical development strategy for DT120 targeting individuals suffering from generalized anxiety disorder (GAD) and major depressive disorder (MDD).
DT120 ODT represents Definium’s primary investigational compound. This pharmaceutical product consists of lysergide tartrate — the tartrate salt variant of LSD — manufactured using Catalent’s proprietary Zydis orally disintegrating tablet platform.
The innovative delivery system aims to enable rapid absorption, enhanced bioavailability, and reduced gastrointestinal adverse reactions when compared to conventional administration routes. The compound functions as a partial agonist targeting serotonin-2A receptors.
DT120 has secured FDA Breakthrough Therapy Designation. Presently, the company is conducting four Phase 3 clinical studies, which represent pivotal milestones for potential commercial approval and market launch.
Stifel Launches Coverage With Bullish Outlook
Just 48 hours prior to the White House policy announcement, Stifel analysts commenced coverage on DFTX stock with a Buy recommendation and established a $30.00 price objective on April 17. With shares hovering around $22.46 during that timeframe, the analyst target suggests potential appreciation of approximately 34%.
This coverage initiation signals expanding institutional attention toward the psychedelic therapeutics sector as policy tailwinds strengthen.
Some Red Flags to Consider
However, not all indicators trend positively. Definium’s GF Score registers a modest 38 out of 100, while profitability metrics score just 1 out of 10 — characteristics typical of early-stage, pre-commercial biotechnology companies without revenue generation.
Financial strength metrics fare considerably better at 7 out of 10, indicating adequate balance sheet positioning. Momentum indicators score 6, consistent with recent trading patterns.
Insider transaction data from the past three months presents a somewhat concerning picture. Corporate insiders completed no stock purchases during this period, while executing sales totaling $0.8 million. While such selling activity isn’t necessarily alarming for clinical-stage biotechnology firms, it warrants investor awareness.
The company currently maintains a market capitalization of approximately $2.24 billion.
The presidential directive dovetails with Definium’s established clinical development strategy, and company leadership expressed enthusiasm for ongoing partnerships with government agencies, healthcare providers, and patient advocacy organizations.
Definium’s DT120 candidate is undergoing clinical assessment for GAD, MDD, and additional serious neurological conditions. The biotechnology firm maintains its headquarters in New York and lists its shares on Nasdaq.



